 
The hydrogen atoms are represented by a circle, shaded grey and labelled 'H'.ġ oxygen atom is connected to 2 hydrogen atoms and the connection is shown by an narrow elongated rectangle shaded pale orange. The oxygen atom is represented by a circle, shaded red and labelled 'O'. This is a schematic diagram of a water molecule. The molecular structure of a water molecule is shown in Figure 6. 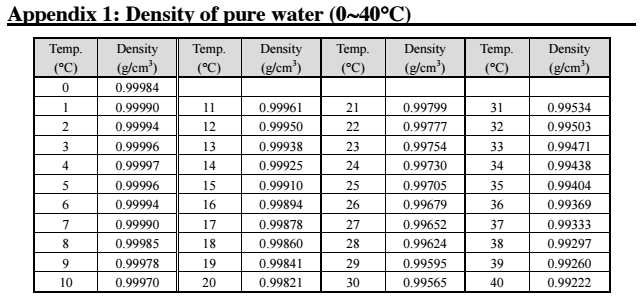
This is an amazing physical property and is why pipes burst and water in cracks shatters rocks in cold temperatures. 
This means that below 3.98 ☌ cooling results in the water molecules spacing out and both the liquid and the solid water below this temperature expand. But cooling a liquid usually packs the molecules more closely, which increases the density. You know from the lake (Figure 5) that ice (i.e. When fresh water at 3.97 ☌ (Figure 4) is cooled the density will decrease. In fresh water the opposite may happen, depending on the temperature. Usually when a liquid is heated the molecules acquire more energy and become more widely spaced, so in the same volume, the density decreases. įigures 5b and c show that the temperature and the density increase with depth from the surface to the bottom layer, where the temperature is 3.98 ☌ and density is at its maximum. 
(b) Temperature structure throughout the lake: there are three layers: a. Figure 5 (a) Section through a lake showing ice on the surface. The green line them runs vertically down through the bottom layer at approximately 999.98 kg m −3. In the intermediate layer the green line shows a linear increase in density with depth from 999.82 kg m −3 to approximately 999.98 kg m −3. The density starts at approximately 999.82 kg m −3 at the surface and remains at that value down to the top of the intermediate layer. The green line shows the same pattern of change as the red temperature line in Figure (b). There is a green line indicating the density. The horizontal axis is located at the top of the figure and is labelled 'density/kg m −3' and is marked from 999.8 kg m −3 to 1000 kg m −3 in intervals of 0.1 kg m −3. It has the same colour and layering as Figure (a), except there are no labels and the top 2 layers (ice and surface layer at 0 ☌) have been merged into one layer. The red line then runs vertically down through the bottom layer at approximately 4 ☌.įigure (c), on the right presents density data as a line graph. In the intermediate layer the red line shows an linear increase in temperature with depth from 0 ☌ to approximately 4 ☌. The red line runs vertically, at a temperature of 0 ☌ from the top of the graph to the top of the intermediate layer. There is a red line indicating the temperature. There is black directional arrow head pointing downward at the lower end of the axis. The vertical axis is labelled 'depth increasing' with no quantitative markings. The horizontal axis is located at the top of the figure and is labelled 'temperature/☌' and is marked from −1 ☌ to 5 ☌ in intervals of 1 ☌. The layer at the bottom is labelled 'deep layer at 3.98 ☌', is the widest of the layers and is shaded in the darker blue.įigure (b), in the middle, presents temperature data as a line graph. The layer below that is labelled 'intermediate layer', is the second narrowest of the 4 layers and is shaded in slightly darker shade of blue than the top 2 layers. The layer below this is labelled 'surface layer at 0 ☌', is the third narrowest of the 4 layers and shaded very pale blue. The top layer is labelled 'ice', is the narrowest of the 4 layers and is shaded very pale blue. This diagram shows the depth profile of the lake along with 4 identified layers to this profile. Trending Questions Why did mendeleev not include germanium in the periodic table? What proccess does a seed start to grow? What is a subscript and where is it located in a chemical formula? What do you mean by the cerebrum controls your brain? Which memorial has two adjoining black granite walls? Is it possible for a Faraday cage to interdict a gamma pulse? Why does quenching in oil produce different results than quenching in water? How many atoms of phosphorus are in a 0.This is a series of 3 diagrams that show the change in temperature and density with depth in a lake and within designated layers within the depth profile of the lake.įigure (a) is on the far left and is labelled 'side view of lake' 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
